Research
Our research aims to identify viral and host factors that regulate infection, replication, and pathogenesis of pandemic-related RNA and DNA viruses. By combining molecular virology, functional genomics, high-throughput screening technologies, and physiologically relevant infection models, we investigate the mechanisms that govern virus-host interactions and explore host-directed antiviral strategies.
In addition to emerging RNA viruses, we study human polyomaviruses, including Merkel cell polyomavirus (MCV), the causative agent of Merkel cell carcinoma. These viruses establish lifelong infections and can cause severe disease in immunocompromised individuals.
Our work integrates genetic manipulation of viral genomes, large-scale functional screening approaches, and primary cell and ex vivo infection systems, allowing us to investigate viral infection and host responses in biologically relevant contexts.
1. Host Factors and Broad-Spectrum Antiviral Discovery
A central goal of our research is to identify host factors required for virus life cycle and to develop host-directed antiviral strategies. Targeting host pathways essential for viral infection may reduce the emergence of resistance and enable broad-spectrum antiviral approaches effective against multiple virus families.
To systematically identify these host factors, we perform large-scale functional screens using siRNA libraries, CRISPR/Cas9 and compound collections, combined with automated high-content imaging to quantify viral replication, cytopathic effects, and host responses. Screening experiments are performed using clinical isolates of respiratory viruses, including coronaviruses, influenza viruses, and flaviviruses.
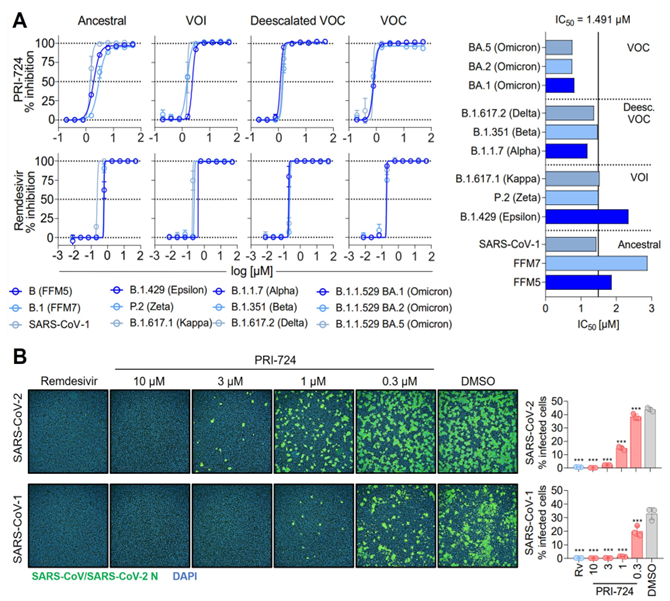
A key strength of our research is the integration of physiologically relevant infection models such as primary human airway epithelial cells cultured at air-liquid interface (ALI), precision-cut lung slices (PCLS) as ex vivo tissue models, primary fibroblasts, and other primary human cell systems into both screening and validation pipelines. In collaboration with partners in APPEAL we combine experimental screening with high-density cell array (HDCA) technology and machine-learning-based data analysis to identify host dependency factors (HDFs), host restriction factors (HRFs), and candidate antiviral compounds.
2. Role of CoV Nucleocapsid Phosphorylation in Virus Life Cycle
As part of the LOEWE-CoroPan consortium, together with Nadine Biedenkopf we investigate the role of coronavirus nucleocapsid protein in virus life cycle.
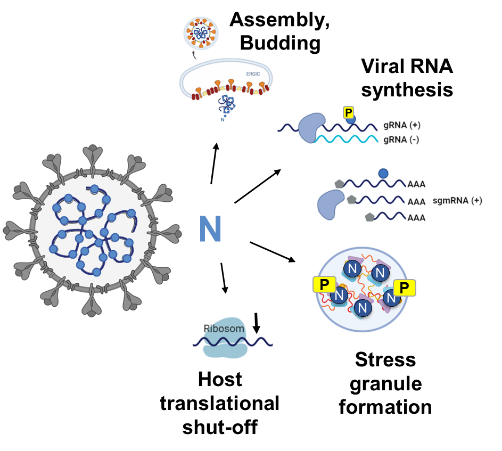
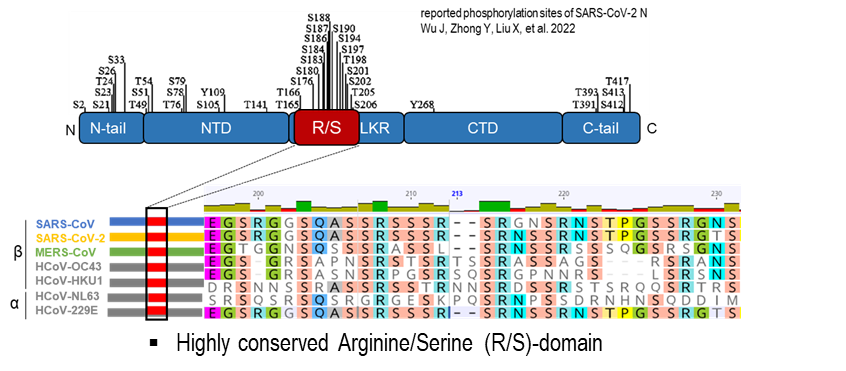
The coronavirus nucleocapsid (N) protein plays a central role in viral RNA synthesis, genome packaging, and transcription of subgenomic mRNAs. Phosphorylation within a conserved arginine-serine-rich (R/S) region is critical for the multifunctional activity of this protein.
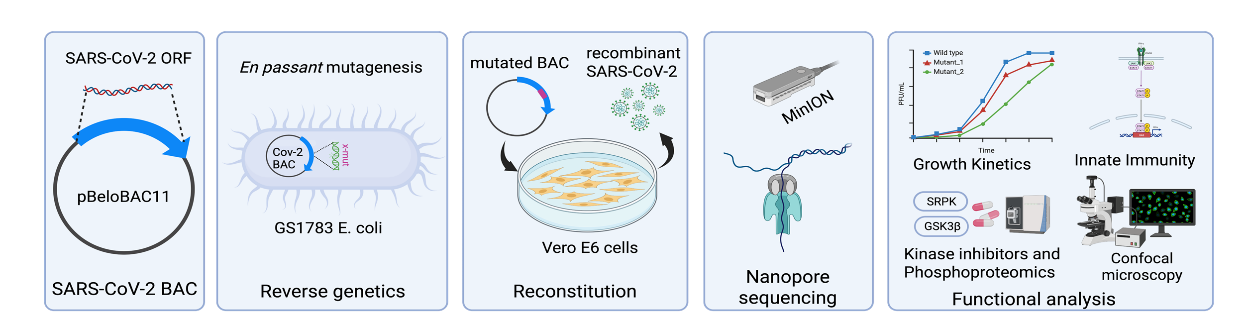
To dissect these regulatory mechanisms, we generate recombinant coronaviruses using bacterial artificial chromosome (BAC) technology. Using en passant mutagenesis, we introduce targeted mutations or reporter genes into viral genomes, enabling precise analysis of how nucleocapsid phosphorylation influences viral replication and transcription.
3. Role of Mixed Lineage Kinase Domain Like Pseudokinase (MLKL) in Virus Infections
In collaboration with Sjoerd van Wijk, we investigate how mixed lineage kinase domain-like pseudokinase (MLKL) regulates viral infection.
MLKL is best known as the executioner of necroptotic cell death, an important innate immune defense mechanism. However, recent evidence suggests that MLKL can also restrict viral replication through non-lytic mechanisms independent of cell death.
Using genetic models, molecular analyses, and advanced infection systems, we study how MLKL influences viral entry, replication, and release from infected cells. By examining both RNA and DNA viruses, we aim to uncover previously unrecognized mechanisms by which host cells control viral infection.
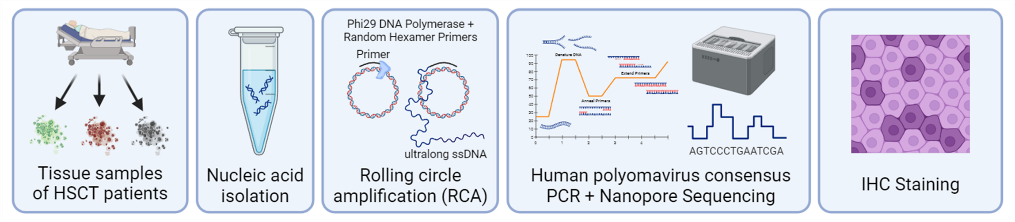
4. Human Polyomavirus-Associated Diseases
Human polyomaviruses are ubiquitous viruses that infect most individuals during early childhood and establish lifelong persistence. Under normal conditions, viral latency is controlled by immune surveillance. However, immunosuppression, such as during organ transplantation or chronic disease, can lead to viral reactivation and disease.
Our research focuses on Merkel cell polyomavirus (MCV), which causes Merkel cell carcinoma, a rare but highly aggressive skin cancer. We also investigate other polyomaviruses associated with inflammatory or non-neoplastic diseases in immunocompromised patients.
Funding
We gratefully acknowledge the support of our funding agencies and partners.